What keeps living things energized?
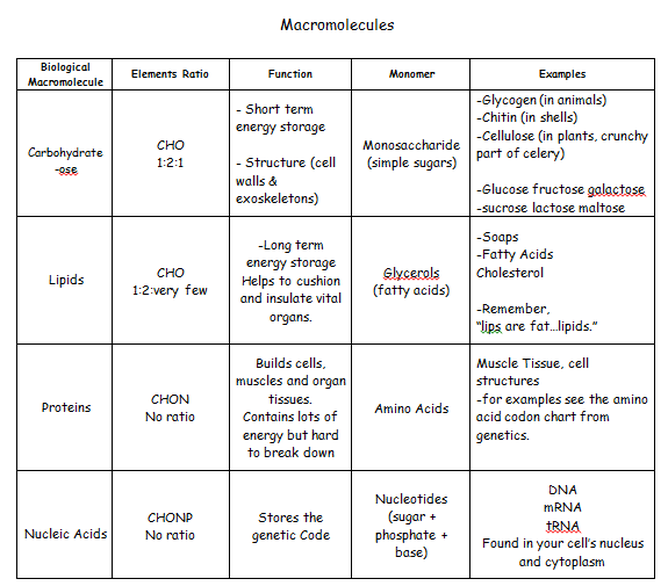
Basics of biomolecules
|
|
|
What is pH?
pH stands for the percent of Hydrogen ions in a substance. These ions make a substance more acidic or basic. The pH scale has a range from 1-14.
Acids have a pH range of 1-6. The 7 range is neutral like water,
and bases have a pH range of 8-14.
Acids are any substance in the range of 1-6. Acids taste sour and tend to be sticky to the touch The lower the pH of an acid the stronger it is!
Example: Orange juice registers around a “4” while battery acid registers around a “1” and can eat through skin.
Bases are any substance in the range of 8-14. Bases tend to be bitter to taste and have a slippery texture, like soap. The higher you go on the scale, the stronger the base!
Example: Baking soda has a pH of about “8” while Bleach (which can eat through clothing) has a pH of about “13”.
pH stands for the percent of Hydrogen ions in a substance. These ions make a substance more acidic or basic. The pH scale has a range from 1-14.
Acids have a pH range of 1-6. The 7 range is neutral like water,
and bases have a pH range of 8-14.
Acids are any substance in the range of 1-6. Acids taste sour and tend to be sticky to the touch The lower the pH of an acid the stronger it is!
Example: Orange juice registers around a “4” while battery acid registers around a “1” and can eat through skin.
Bases are any substance in the range of 8-14. Bases tend to be bitter to taste and have a slippery texture, like soap. The higher you go on the scale, the stronger the base!
Example: Baking soda has a pH of about “8” while Bleach (which can eat through clothing) has a pH of about “13”.